There is an ongoing search for better and more effective treatments for cancer. The reason cancer is difficult to treat is because many cancer cells can evade the immune system. Chimeric antigen receptor (CAR) T cell therapy uses the patient’s own immune cells to treat their cancer. During CAR-T therapy, T cells, which are immune system cells that help kill off pathogens, are extracted from a patient’s blood. These T cells are then mixed with a virus that allows the T cells to develop a chimeric antigen receptor (CAR), an artificial receptor that will navigate toward the location of the cancer. Then, the CAR-T cells are released back into the patient’s bloodstream intravenously. The CAR-T cells track and home in on a tumor-associated antigen (TAA) that is specific to the patient’s cancer. The CAR-T cells then release molecules that trigger cell death pathways within the tumor cells. This process is illustrated in Figure 1.
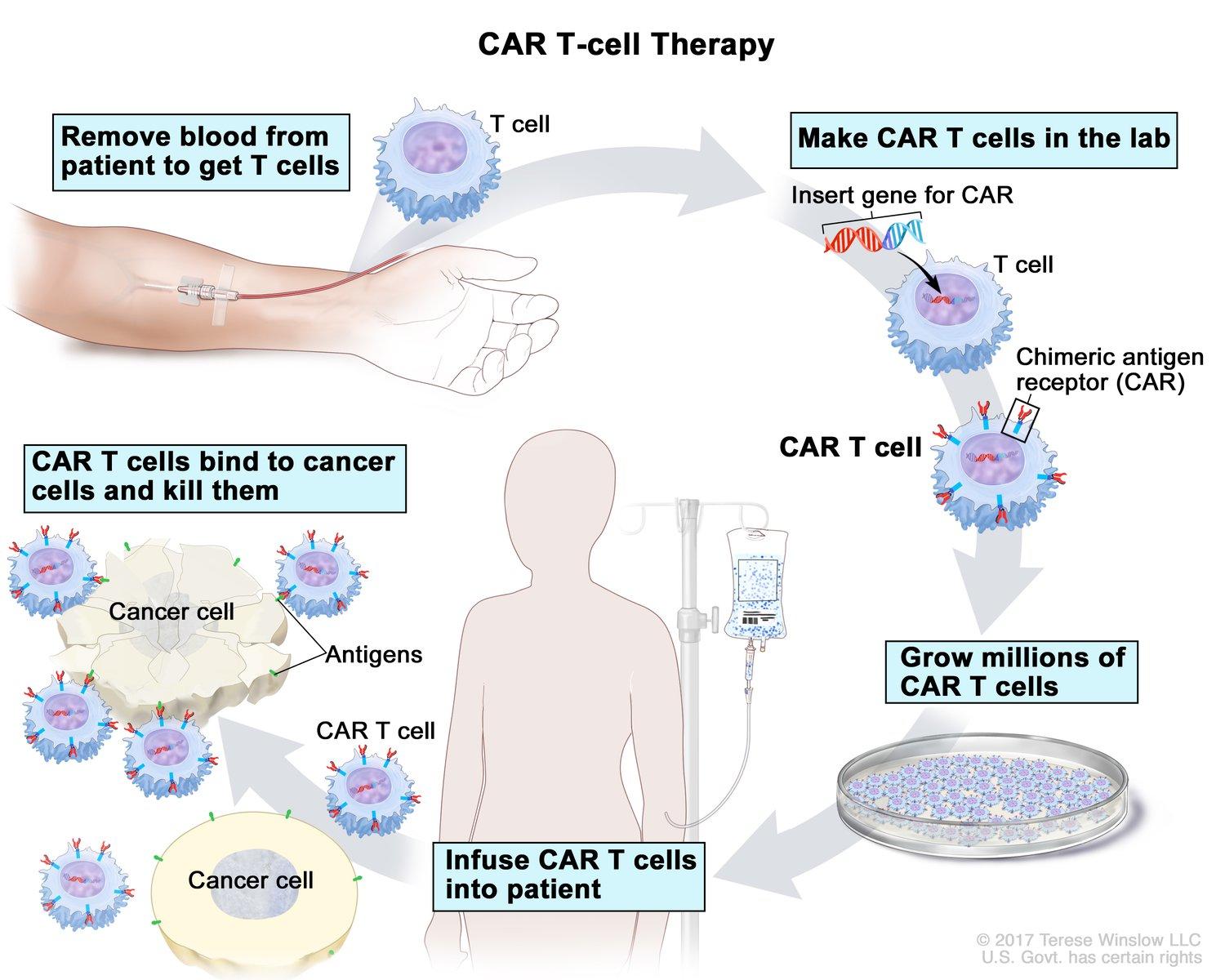
Figure 1 CAR-T cell therapy process from the National Cancer Institute.
CAR-T cells are shown to be effective at treating blood cancers such as leukemia and lymphoma. Despite the success CAR-T cells have with these cancer types, this therapy is much less effective for treating solid tumors. CAR-T cells can only induce lysis once they bind to the cell surface. However, solid tumors have a small surface area to volume ratio, making it difficult for CAR-T cells to infiltrate and reach all the tumor cells. Even if it does, the rate at which the tumor cells are killed by this process is much slower than the rate at which the tumor cells replicate, rendering the system inefficient. Another challenge lies in the identification of markers that are “specifically and uniformly expressed on heterogeneous solid tumors” (Vincent et al., 2023). Heterogeneous solid tumors are ones that have a sub-population of cells with different features to that of the tumor cells. TAAs identified are rarely tumor-specific, meaning most antigens are present in healthy cells too. Thus, the use of these TAAs carries a high risk of fatal on-target, off-tumor toxicity that kills the vital, healthy cells of the patient.
To better treat solid tumors, Vincent et al. from Columbia University took a slightly different approach whereby they created probiotic-guided CAR-T cells (ProCARs) that responded to synthetic CAR targets released by tumor-colonizing colonizing probiotic bacteria. The researchers engineered a probiotic strain, Escherichia coli Nissle 1917 (EcN), which is used as a delivery vesicle for synthetic CAR targets called Tags. EcN is able to colonize tumor cells and grow in tumor microenvironments (TMEs) and contains quorum-regulated circuits, meaning bacteria will die after reaching a certain population density threshold and release Tags within the tumor. When Tags are released, they broadly bind to surface and matrix proteins of the tumor cell. CAR-T cells are engineered with GFP28z receptors, which will bind to Tags and activate the CAR-T cell. Thus, activation of CAR-T cells allows for Tag-mediated in situ lysis of the tumor cells.
Overall, this system overcomes the two previous challenges faced when treating solid tumors. By using Tag, this system allows for antigen-agnostic immunotherapy, meaning eliciting an immune response against tumor cells without the identification of TAAs. Furthermore, since the probiotic bacteria migrates to the tumor location, this treatment mechanism minimizes off-tumor toxicity. Last but not least, the advantage of using bacteria in this system lies in its ability to colonize tumor cells and grow within TMEs, enabling treatment options for difficult-to-infiltrate tumors.
This system has shown significant lysis across seven human cancer cell lines. Empirical data reflect no significant weight loss shown in mice, no bacterial growth outside of tumor cells or in any healthy organs, and Tag plasmids were well maintained in vivo. While this treatment has been shown to be effective, the potential toxicity from repeated dosing of bacterial therapy in humans is still an important concern for clinical translation, since humans are more sensitive to toxins inside the bacterial cell than mice. Overall, the researchers conclude that probiotic-guided CAR-T cells may provide a novel and safe treatment method for solid tumors.
Works Cited:
T-cell Transfer Therapy—Immunotherapy—NCI (nciglobal,ncienterprise). (2022, April 1). [cgvArticle]. https://www.cancer.gov/about-cancer/treatment/types/immunotherapy/t-cell-transfer-therapy
Vincent, R. L., Gurbatri, C. R., Li, F., Vardoshvili, A., Coker, C., Im, J., Ballister, E. R., Rouanne, M., Savage, T., de los Santos-Alexis, K., Redenti, A., Brockmann, L., Komaranchath, M., Arpaia, N., & Danino, T. (2023). Probiotic-guided CAR-T cells for solid tumor targeting. Science, 382(6667), 211–218. https://doi.org/10.1126/science.add7034