Context
Marine phytoplankton are microscopic algae integral to oceanic ecosystems and global biogeochemical cycles. They contribute significantly to the process of carbon displacement into the deep ocean and primary production, forming the foundation of the marine food web. However, these phytoplankton populations are vulnerable to various environmental and biological stressors, including temperature changes, nutrient availability, and pathogen infections. When a phytoplankton bloom collapses, bacteria consume their organic matter, a process that requires oxygen. The decay of the bloom and oxygen levels can deplete oxygen and lead to “dead zones” that can suffocate marine life (US EPA, 2013). Researching the components of bloom dynamics enables us to better understand their interactions as a foundation of the food web and regulator of oxygen levels.

Fig 1. Demise of a phytoplankton bloom over the course of a handful of days (Demise of a Phytoplankton Bloom, 2014)
One of the most abundant bloom-forming phytoplankton is Gephyrocapsa Huxley, a species of coccolithophore, a type of phytoplankton covered in calcium carbonate plates known for its widespread blooms in the ocean. While viral infections have long been recognized as a primary cause of phytoplankton bloom collapse, researchers have questioned whether bacteria could be another potential source of pathogenicity. Recent research regarding G. huxleyi phytoplankton suggests so.

Fig 2. Calcium carbonate plating on a coccolithophore phytoplankton (Briggs, 2021)
This groundbreaking study by Lang-Yona et al. investigated whether airborne bacteria could infect G. huxleyi blooms and be an explanation for bloom collapses. This research aimed to analyze atmospheric bacteria as an ecological regulator of phytoplankton populations, an often disregarded consideration in the dynamics of oceanic microbial interactions and climate models. Understanding these interactions is critical for predicting changes in marine ecosystems and their impact on global carbon cycles.
Methods of the Study
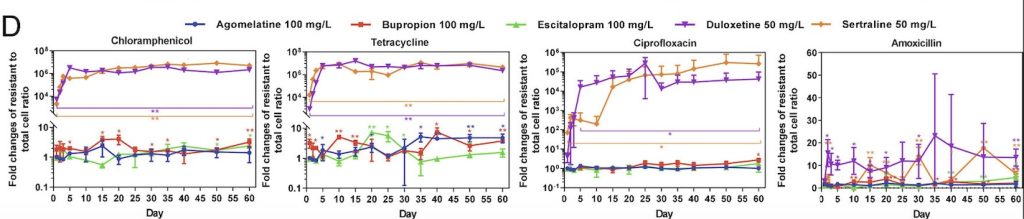
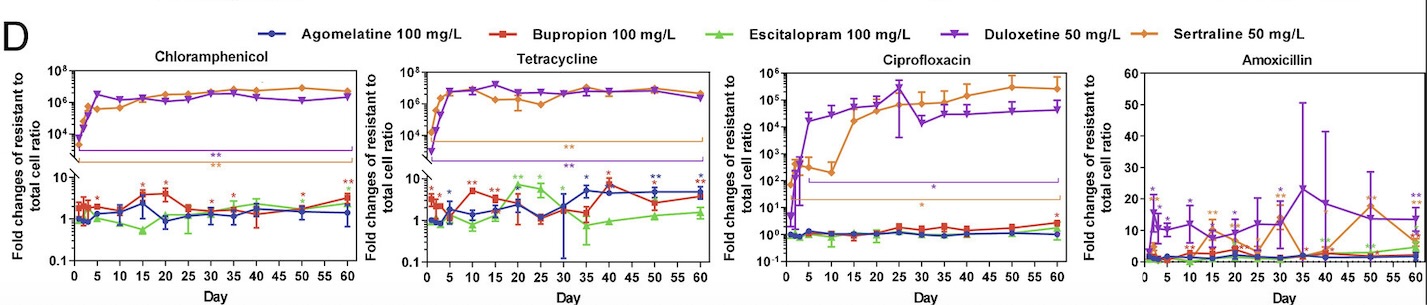
To explore whether airborne bacteria play a role in controlling phytoplankton populations, researchers collected air and water samples above a bloom of G. huxleyi in the North Atlantic. They conducted this work aboard the research vessel R/V Tara, using specialized instruments to capture airborne bacteria at different heights. These included high-volume air samplers and devices called cascade impactors, which were set up at different points on the ship, including the deck and mast. This setup allowed them to collect bacteria from various altitudes and better understand how microbes travel through the air (Lang-Yona et al., 2024)
Back in the lab, the team introduced the airborne bacteria into cultures of G. huxleyi to see what would happen. They carefully watched for signs of infection, such as a drop in the algae’s natural fluorescence (a sign they were losing their ability to photosynthesize), increased debris in the water (indicating cell death), and visible damage to the algal cells. When signs of infection appeared, they filtered out the bacteria from the cultures and grew them on a nutrient-rich surface called Marine Agar 2216. This step helped them isolate specific bacterial strains. To confirm that these bacteria were truly responsible for the infection, they then reintroduced them to fresh G. huxleyi cultures and checked whether the same effects occurred.
To track how the bacteria and algae interacted over time, researchers used a technique called flow cytometry. This method shines a laser through tiny droplets of water containing cells, measuring their size, shape, and natural glow. It allows scientists to quickly count how many algae and bacteria are present and determine how the infection is progressing.
Finally, they identified the bacteria using genetic sequencing and measured their presence in air and water samples with a technique called quantitative PCR (qPCR). This method detects and counts bacterial DNA, helping researchers understand how common these airborne microbes are in different environments.
Results of the Study and Implications
The study identified the airborne bacterium Roseovarius nubinhibens as a key bacteria capable of infecting and contributing to the collapse of G. huxleyi blooms. This bacterium was found to remain viable after atmospheric transport and effectively infects phytoplankton upon reaching ocean waters. The ability of R. nubinhibens to survive and remain pathogenic after airborne dispersal suggests a more dynamic role for bacteria in ocean-atmosphere interactions than previously recognized.
The ability of bacteria to be transported via wind patterns indicates a geologically vast and major mechanism that has previously been overlooked. The findings suggest that in addition to viral infections, bacterial pathogens may serve as natural regulators of phytoplankton populations, influencing bloom duration and oceanic carbon cycling. This discovery is particularly significant because phytoplankton blooms play a critical role in the global carbon cycle by taking carbon from the atmosphere, and upon death, sinking to the deep ocean where the carbon is stored. If bacterial infections contribute to bloom collapse and can be dispersed so vastly, they may influence carbon fluxes in ways that need to be accounted for in climate models.
Previously, viral infections were considered the primary biological driver of bloom decline, but this study introduces airborne bacteria as an additional player in phytoplankton mortality. This raises important questions about how environmental factors such as wind patterns and ocean currents influence bacterial dispersal. Additionally, climate change may impact the spread of airborne pathogens, potentially altering bloom dynamics in unforeseen ways. A warming climate and shifting atmospheric circulation patterns could enhance or suppress the spread of algicidal bacteria, with cascading effects on marine ecosystems.
Furthermore, this research highlights the complexity of microbial interactions in the ocean. Many bacterial species exhibit “Jekyll-and-Hyde” dynamics, shifting between mutualism and pathogenicity depending on environmental conditions and the physiological state of their algal hosts. In the case of R. nubinhibens, it is possible that under certain conditions, it exists in a neutral or even beneficial relationship with G. huxleyi, but when environmental factors such as nutrient depletion or increased bacterial density trigger a shift, it becomes pathogenic. The study’s infection experiments demonstrated that R. nubinhibens could rapidly induce algal demise, suggesting a transition from a benign to an algicidal state. This aligns with previous findings that some marine bacteria can switch between cooperative and harmful interactions based on chemical signaling. Understanding these complex interactions is essential for developing a more accurate picture of microbial regulation in marine environments, as such shifts can significantly alter bloom dynamics and oceanic food webs.
This study provides new insight into the role of airborne bacteria in regulating marine phytoplankton populations, demonstrating that Roseovarius nubinhibens can contribute to G. huxleyi bloom collapse. These findings expand our understanding of ocean-atmosphere microbial interactions and introduce airborne bacteria as an important but previously overlooked factor in bloom dynamics. Incorporating airborne bacterial processes into ecological and climate models will be crucial for accurately predicting future oceanic changes. Further research is necessary to determine whether other phytoplankton species are similarly affected and how environmental shifts may influence the prevalence and impact of airborne bacterial infections on marine ecosystems. Understanding these dynamics is essential for assessing ocean health and resilience in a rapidly changing climate.
References: