Cognitive decline with age is a major concern in medicine and public health. In 2021, the World Health Organization reported 57 million people were affected by dementia worldwide (World Health Organization, 2023). Well-established risk factors include alcohol intake, lower education level, physical inactivity, obesity, and diabetes, and preventive strategies have developed steadily. However, one potential contributor is often overlooked in major dementia research: tooth loss. Galindo-Moreno et al. (2022) examined this relationship through a large-scale analysis of over 100,000 US Americans, making a case for oral health as an underrecognized factor in cognitive decline.
Edentulism refers to the partial or complete loss of permanent teeth. Edentulism can be caused by a multitude of factors, including biological processes such as caries (tooth decay) and periodontal disease (infection or inflammation of gums and bone), pulpal pathologies (damage to nerves, tissue, and blood vessels in the center of a tooth), trauma, or oral cancer. In addition to biological causes, edentulism can result from factors affecting dental care: patient preference, access to care, treatment options, and health insurance (Felton 2009). A study found 37% of edentulism cases were due to extraction from caries, 29% from periodontal diseases, and 12% due to trauma (Al-Rafee 2020).
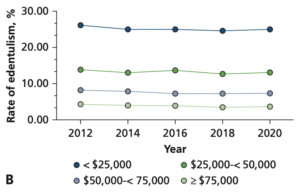
Although oral health care has developed significantly in the last few decades, edentulism remains a prevalent and irreversible condition (Al-Rafee 2020). It can occur at all ages, but the highest incidence occurs between the ages of 75-79 [Figure 1] (Chen et al. 2025). Those most affected by tooth loss typically have a lower socioeconomic standing, which makes health care less affordable and accessible [Figure 2] (Vemulapalli et al. 2024)

Galindo-Moreno et al. proposed multiple pathways by which tooth loss can lead to cognitive decline. Two that play directly into known factors are the “diet and nutrition mechanism” and the masticatory mechanism. The number of teeth and which teeth are present affect what we can eat and how we eat. Mastication — chewing of food (Xu et al. 2008) — is directly influenced by edentulism due to the reduced bite force one can exert with missing teeth or dentures (Galindo-Moreno et al. 2022; Weijenberg et al. 2011). Changes to mastication may impact cognition by decreasing sensory input, which would reduce cell growth and development, impairing the cholinergic neurotransmitter system responsible for regulating memory, muscles, and attention, and reducing the generation of new neurons triggered by exercise (Weijenberg et al. 2011). Mastication additionally restricts our diet and therefore directly plays into the diet and nutrition mechanism. Often, with altered dentition, chewing can be an immense hurdle, for which the solution is a softer yet less nutritious diet.Nutrients such as omega-3 fatty acids, B vitamins, and antioxidants have important neuroprotective properties that help preserve the blood brain barrier, an essential layer that prevents toxins from entering the brain,additionally reducing inflammation, lowering the risk of cognitive decline (Power et al. 2019). Both the masticatory and diet and nutrition mechanisms are intertwined with diabetes and obesity, which are known risk factors for cognitive decline (Galindo-Moreno et al. 2022).
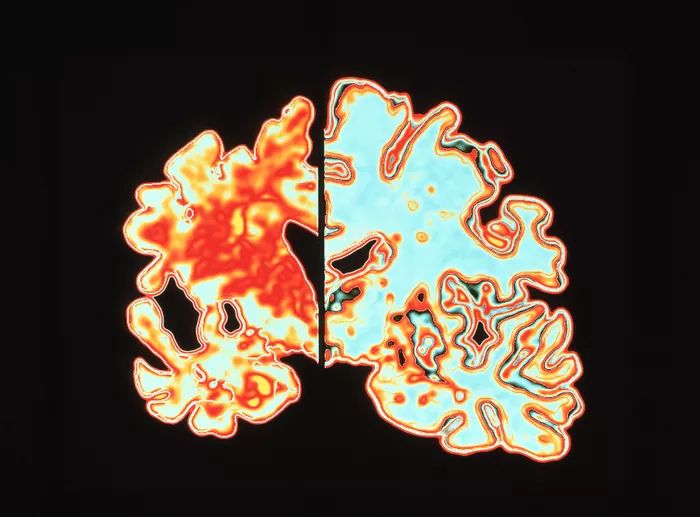
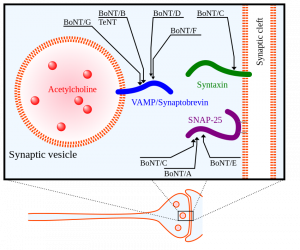
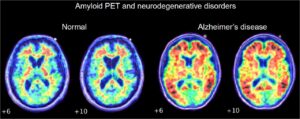
Another pathway this study mentions is the inflammation/infection mechanism. A leading cause of edentulism is periodontitis, a severe gum infection often driven by the bacterium Porphyromonas gingivalis. This bacterium induces the local release of cytokines, proinflammatory proteins (Galindo-Moreno et al. 2022). Once in the bloodstream, cytokines promote the production of amyloid-β, a peptide whose accumulation is associated with Alzheimer’s disease (Leira et al. 2020). Simultaneously, Porphyromonas gingivalis increases the permeability of the blood-brain barrier (Lei et al. 2023). The heightened permeability of the BBB causes accumulation of overproduced amyloid-β in the brain tissue [Figure 3] (Galindo-Moreno et al. 2022; Leira et al. 2020)
To investigate the relationship between tooth loss and cognitive decline, the researchers analyzed data from over 100,000 Americans drawn from two large national health surveys, NHIS (2014-2017) and NHANES (2005-2018). The NHIS survey was particularly well-suited for assessing cognitive state, as it included four questions on concentration and memory. However, the survey included only one binary dental question asking whether the participants had a complete dentition or had lost ≥1 teeth. The NHANES survey complemented this with a thorough section on dental records. The exact number and location of lost teeth were documented. However, it assessed cognitive state with only one question on memory and confusion (Galindo-Moreno et al. 2022).
Their primary statistical tool was multinomial logistic regression, a method used when an outcome has more than two categories. In this case, the categories were cognitive difficulty, ranging from “none” to “some” to “a lot.” By using this model, the researchers simultaneously accounted for other factors known to affect cognitive health, including age, income, education level, depression, anxiety, cardiovascular health, and lifestyle habits such as smoking and exercise, which were included in the health surveys. By modeling these variables together, the researchers could estimate the independent contribution of tooth loss to cognitive decline.
The results were expressed as odds ratios (ORs), which indicate how much more likely a given outcome is in one group than in a reference group. Here, the reference was a fully toothed person reporting no cognitive difficulties. An OR above 1.0 indicated higher odds of cognitive problems among people with missing teeth. This held true even after the other variables were statistically accounted for. The researchers also used a technique called ROC curve analysis on the NHANES data that included exact tooth counts, allowing them to identify a meaningful threshold below which cognitive risk measurably increased (Galindo-Moreno et al. 2022).
The researchers found that, overall, the presence of teeth was statistically associated with a better cognitive state. The NHIS data showed that people with edentulism (partial or complete) had an OR > 1 across all cognitive categories, especially memory, even after accounting for other risk factors. This trend was also observed across categories of gender, socio-economic status (SES), education, and cardiovascular risk — all of which negatively impact cognition. Notably, socioeconomic status emerged as one of the strongest predictors, alongside edentulism, reflecting how directly financial circumstances shape access to dental care and, through it, long-term cognitive health.
Using ROC curve analysis of the NHANES data, they determined the threshold for cognitive risk to be 20.5 teeth, indicating that a person with fewer than 21 teeth has an increased risk of cognitive decline compared to a fully dentulous person (Galindo-Moreno et al. 2022). Importantly, the study analyzed the NHANES survey and found a gradient effect: the fewer teeth a person had, the worse their cognitive outcomes tended to be, which strengthens the case that the association is meaningful rather than coincidental. Furthermore, a threshold could be determined for each individual tooth category: 5.5, 5.5, 3.5, 4.5, respectively, for molars, premolars, canines, and incisors. The multinomial regression of the NHANES data determined molars had the highest OR. The researchers linked this to the masseter, an important masticatory muscle supported by molars, which may, through its activity, stimulate the release of neurotrophic factors that support brain health.
The link between edentulism and cognitive decline is still scarcely researched. As of March 2026, there are only 66 results on PubMed, 142 on ScienceDirect, and 148 on Wiley on the correlation between edentulism and cognitive decline. To put this into perspective, there are 2,277 results on PubMed, 18,967 on ScienceDirect, and 10,546 on Wiley on the relationship between diet and cognitive decline. The discussed research article combines two USA national health surveys with diverse samples, NHIS and NHANES, making it one of the largest in scope to date on tooth loss and cognitive decline. Although Galindo-Moreno and his team compellingly demonstrate the correlation, they recognize that their findings cannot answer whether edentulism leads to poorer cognition or rather poor cognition leads to edentulism (Galindo-Moreno et al. 2022, 3498). Some of the issues the researchers faced were the binary assessment of dentition in the NHIS survey, the single question on cognitive condition in the NHANES survey, and the overall lack of records on the reasons for tooth loss (Galindo-Moreno et al. 2022).
Nevertheless, this study is a step in the right direction. Galindo-Moreno et al. showed that edentulism is correlated with cognition, thereby providing meaningful epidemiological evidence for a relatively young field. Consequently, this study and further research could have great clinical implications for cognitive health, not only in cost-effective treatment and prevention, but also in an important personal impact for those struggling with cognitive impairments and dental hygiene.
Al-Rafee, Mohammed A. 2020. “The Epidemiology of Edentulism and the Associated Factors: A Literature Review.” Journal of Family Medicine and Primary Care 9 (4): 1841–43. https://doi.org/10.4103/jfmpc.jfmpc_1181_19.
Chapleau, Marianne, Leonardo Iaccarino, David Soleimani-Meigooni, and Gil D. Rabinovici. 2022. “The Role of Amyloid PET in Imaging Neurodegenerative Disorders: A Review.” Clinical Investigation. Journal of Nuclear Medicine63 (Supplement 1): 13S-19S. https://doi.org/10.2967/jnumed.121.263195.
Chen, Hui Min, Kuo Shen, Ling Ji, Colman McGrath, and Hui Chen. 2025. “Global and Regional Patterns in Edentulism (1990-2021) With Predictions to 2040.” International Dental Journal 75 (2): 735–43. https://doi.org/10.1016/j.identj.2024.11.022.
“Dementia.” n.d. Accessed March 27, 2026. https://www.who.int/news-room/fact-sheets/detail/dementia.
Felton, David A. 2009. “Edentulism and Comorbid Factors.” Journal of Prosthodontics 18 (2): 88–96. https://doi.org/10.1111/j.1532-849X.2009.00437.x.
Galindo-Moreno, Pablo, Lucia Lopez-Chaichio, Miguel Padial-Molina, et al. 2022. “The Impact of Tooth Loss on Cognitive Function.” Clinical Oral Investigations 26 (4): 3493–500. https://doi.org/10.1007/s00784-021-04318-4.
Lei, Shuang, Jian Li, Jingjun Yu, et al. 2023. “Porphyromonas Gingivalis Bacteremia Increases the Permeability of the Blood-Brain Barrier via the Mfsd2a/Caveolin-1 Mediated Transcytosis Pathway.” International Journal of Oral Science15 (January): 3. https://doi.org/10.1038/s41368-022-00215-y.
Leira, Yago, Álvaro Carballo, Marco Orlandi, et al. 2020. “Periodontitis and Systemic Markers of Neurodegeneration: A Case–Control Study.” Journal of Clinical Periodontology 47 (5): 561–71. https://doi.org/10.1111/jcpe.13267.
Power, Rebecca, Alfonso Prado-Cabrero, Ríona Mulcahy, Alan Howard, and John M. Nolan. 2019. “The Role of Nutrition for the Aging Population: Implications for Cognition and Alzheimer’s Disease.” Annual Review of Food Science and Technology 10 (1): 619–39. https://doi.org/10.1146/annurev-food-030216-030125.
Vemulapalli, Abhilash, Surendra Reddy Mandapati, Anusha Kotha, Hemanth Rudraraju, and Subhash Aryal. 2024. “Prevalence of Complete Edentulism among US Adults 65 Years and Older.” The Journal of the American Dental Association 155 (5): 399–408. https://doi.org/10.1016/j.adaj.2024.02.002.
Weijenberg, R. A. F., E. J. A. Scherder, and F. Lobbezoo. 2011. “Mastication for the Mind—The Relationship between Mastication and Cognition in Ageing and Dementia.” Neuroscience & Biobehavioral Reviews 35 (3): 483–97. https://doi.org/10.1016/j.neubiorev.2010.06.002.
World Health Organization. 2023. “Dementia” Fact Sheets. https://www.who.int/news-room/fact-sheets/detail/dementia
Xu, W. L., J. E. Bronlund, J. Potgieter, et al. 2008. “Review of the Human Masticatory System and Masticatory Robotics.” Mechanism and Machine Theory 43 (11): 1353–75. https://doi.org/10.1016/j.mechmachtheory.2008.06.003.